SLC22A6 Antikörper
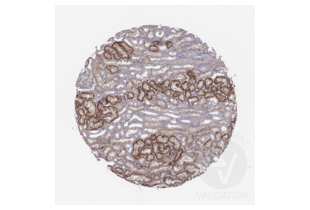
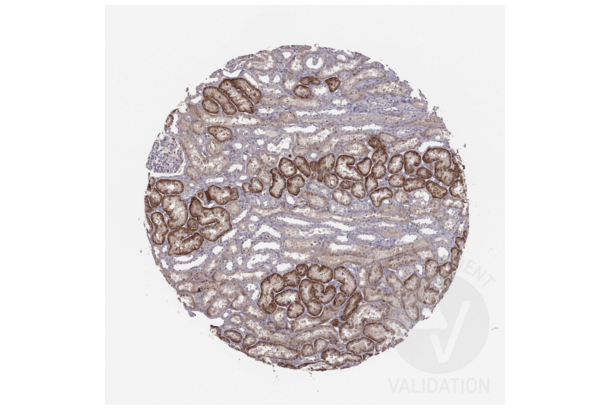 SLC22A6 Antikörper (C-Term) (ABIN3044261)
SLC22A6 Antikörper (C-Term) (ABIN3044261)
SLC22A6 Reaktivität: Human, Ratte WB, IHC (p) Wirt: Kaninchen Polyclonal unconjugated
SLC22A6 Reaktivität: Human WB, FACS, IHC (p) Wirt: Kaninchen Polyclonal RB23819 unconjugated
SLC22A6 Reaktivität: Human, Ratte WB, IHC, ELISA Wirt: Kaninchen Polyclonal unconjugated
SLC22A6 Antikörper nach Reaktivität
Hier sind SLC22A6 Antikörper für eine Vielzahl von Species wie anti-Human SLC22A6, anti-Rat SLC22A6, anti-Mouse SLC22A6 zu finden. Die unten aufgeführten Species gehören zu den verfügbaren Arten. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
SLC22A6 Antikörper nach Anwendung
Hier sind SLC22A6 Antikörper zu finden, welche für eine bestimmte Anwendung wie WB, IHC, ELISA, FACS validiert wurde. Einige der verfügbaren Anwendungen sind unten aufgeführt. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
SLC22A6 Antikörper nach Wirt
Hier sind SLC22A6 Antikörper mit einem spezifischen Wirt zu finden. Die hier aufgeführten Wirt sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
SLC22A6 Antikörper nach Bindungsspezifität
Hier sind SLC22A6 Antikörper mit einem bestimmten Epitop aufgelistet. Die unten aufgeführten Epitope gehören zu den verfügbaren Epitopen. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
SLC22A6 Antikörper nach Klonalität
Finden Sie verfügbare monoklonale oder polyklonale SLC22A6 Antikörper. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
SLC22A6 Antikörper nach Klon
Hier sind SLC22A6 Antikörper mit einem spezifischen Klon zu finden. Die hier aufgeführten Klon sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
SLC22A6 Antikörper nach Konjugat
Suchen Sie SLC22A6 Antikörper mit einer bestimmten Konjugation wie Biotin, Alexa Fluor 488, Alexa Fluor 555. Die unten aufgeführten Konjugate gehören zu den verfügbaren. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
Häufig verwendete SLC22A6 Antikörper
- (2)
- (1)
- (1)
- (3)
- (3)
- (3)
- (3)
- (3)
- (2)
- (3)
- (1)
- (1)
- (1)
Aktuelle Publikationen für unsere SLC22A6 Antikörper
: "Phloretin attenuates hyperuricemia-induced endothelial dysfunction through co-inhibiting inflammation and GLUT9-mediated uric acid uptake." in: Journal of cellular and molecular medicine, Vol. 21, Issue 10, pp. 2553-2562, (2018) (PubMed).: "Perfluorononanoic acid in combination with 14 chemicals exerts low-dose mixture effects in rats." in: Archives of toxicology, Vol. 90, Issue 3, pp. 661-75, (2016) (PubMed).
: "The multispecific organic anion transporter (OAT) family." in: Pflügers Archiv : European journal of physiology, Vol. 440, Issue 3, pp. 337-50, (2000) (PubMed).
: "[Molecular mechanisms of drug transport]." in: Nihon yakurigaku zasshi. Folia pharmacologica Japonica, Vol. 116, Issue 3, pp. 114-24, (2000) (PubMed).
Aliase für SLC22A6 Antikörper
solute carrier family 22 member 6 (SLC22A6) Antikörpersolute carrier family 22 (organic anion transporter), member 6 (Slc22a6) Antikörper
solute carrier family 22 (organic anion transporter), member 6, like (slc22a6l) Antikörper
Solute carrier family 22 member 6 (s22a6) Antikörper
solute carrier family 22 member 6 (Slc22a6) Antikörper
solute carrier family 22 (organic anion transporter), member 6 S homeolog (slc22a6.S) Antikörper
HOAT1 Antikörper
mOat1 Antikörper
NKT Antikörper
nkt Antikörper
OAT1 Antikörper
Oat1 Antikörper
oat1 Antikörper
oat1-B Antikörper
Orctl1 Antikörper
PAHT Antikörper
Paht Antikörper
paht Antikörper
ROAT1 Antikörper
Roat1 Antikörper
roat1 Antikörper
slc22a6 Antikörper
SLC22A6 Antikörper
Slc22a6 Antikörper
slc22a6-B Antikörper
zgc:77073 Antikörper
Haben Sie etwas anderes gesucht?
- SLC22A5 Antikörper
- SLC22A4 Antikörper
- SLC22A3 Antikörper
- SLC22A25 Antikörper
- SLC22A24 Antikörper
- SLC22A23 Antikörper
- SLC22A2 Antikörper
- SLC22A17 Antikörper
- SLC22A16 Antikörper
- SLC22A15 Antikörper
- SLC22A14 Antikörper
- SLC22A13 Antikörper
- SLC22A12 Antikörper
- SLC22A11 Antikörper
- SLC22A1 Antikörper
- SLC20A2 Antikörper
- SLC20A1 Antikörper
- SLC1A7 Antikörper
- SLC1A6 Antikörper
- SLC1A5 Antikörper
- SLC22A8 Antikörper
- SLC22A9 Antikörper
- SLC23A1 Antikörper
- SLC23A2 Antikörper
- SLC23A3 Antikörper
- SLC24A1 Antikörper
- SLC24A2 Antikörper
- SLC24A3 Antikörper
- SLC24A4 Antikörper
- SLC24A5 Antikörper
- SLC24A6 Antikörper
- Slc25a1 Antikörper
- SLC25A10 Antikörper
- SLC25A11 Antikörper
- SLC25A12 Antikörper
- slc25a13 Antikörper
- SLC25A14 Antikörper
- SLC25A15 Antikörper
- SLC25A16 Antikörper
- SLC25A17 Antikörper




