Rekombinanter SARS-CoV-2 Spike S1 Antikörper (RBD)
Kurzübersicht für Rekombinanter SARS-CoV-2 Spike S1 Antikörper (RBD) (ABIN6952546)
Target
Alle SARS-CoV-2 Spike S1 Antikörper anzeigenAntikörpertyp
Reaktivität
Wirt
Klonalität
Konjugat
Applikation
Klon
-
-
Highlights
-
- Recombinant human neutralizing antibody (hNAb) against SARS-CoV-2.
- Binds SARS-CoV-2 S proteins of lineages B.1.1.7 (alpha), B.1.351 (beta), P.1 (gamma), B.1.617.2 (delta), B.1.1.529 (omicron), B.1.429 (epsilon), B.1.525 (eta), and B.1.617.1 (kappa).
- Frequently used as reference in S-protein ELISAs and neutralization assays.
- Synergizes with other hNAbs: binds a highly conserved epitope, not interefering with the S-protein's ACE2 RBD.
-
Bindungsspezifität
- RBD
-
Verwendungszweck
- Recombinant monoclonal antibody CR3022 to SARS-CoV S Glycoprotein.
-
Spezifität
-
The antibody CR3022 binds the amino acids 318-510 in the S1 domain of the SARS-CoV Spike protein as well as SARS-CoV-2 (COVID-19) Spike protein. The antibody also binds to P462L-substituted S318-510 fragments of the SARS spike protein. The binding epitope is only accessible in the "open" conformation of the spike protein (Joyce et al. 2020).
While most S-protein RBD binding antibodies compete for antigen binding with ACE2, the CR3022 epitope does not overlap with the ACE2-binding site. It does thus not hinder binding of neutralizing antibodies. While CR3022 on its own exhibits only a weak neutralizing effect, it has been shown to synergize with other S-protein RBD binding antibodies to neutralize SARS-CoV. This effect still has to be confirmed in context with SARS-CoV-2 (Yuan et al. 2020).
-
Kreuzreaktivität (Details)
- The anti-SARS-CoV-2 antibody CR3022 was originally discovered in a SARS patient, but it was shown to be a potent binder of SARS-CoV-2 spike protein (S1).
-
Produktmerkmale
-
Original Species of Ab: Human
Original Format of Ab: IgG1
-
Aufreinigung
- Protein A affinity purified
-
Immunogen
- The original monoclonal antibody was generated through an scFv library derived from a peripheral blood lymphocytes of a patient exposed to the SARS-CoV.
-
Isotyp
- IgG1 kappa
-
-
-
-
Applikationshinweise
- This antibody binds to both SARS-CoV and SARS-CoV-2 with high affinity (PMID: 16796401 & 32065055). The initial characterization of the binding of this antibody was performed by ELISA and indicates potential for the development of diagnostic assays, as both virus-capture assays, or as controls in serological assays measuring immune-responses to virus exposure. Human IgG1, IgG3, IgM and IgA isotypes are available to mimic antibody responses seen in COVID19 < a href="https://www.medrxiv.org/content/10.1101/2020.03.17.20037713v1" target="_blank">(Amanat et al. 2020). Human IgG2 and IgG4 subtypes, which are also seen in a small subset of COVID-19 patients, are also available to investigate their role in the response to SARS-CoV-2. The original human IgG1 version of the antibody works synergistically in combination with another non-ompeting SARS antibody CR3014 and is a potential candidate for passive immune prophylaxis of SARS-CoV infection (Meulen et al., 2006). The original antibody (human IgG1) was also reported to bind the 2019-nCoV RBD (KD of 6.3 nM). This antibody has been attributed a potential to be developed as a therapeutic agent, alone or in combination with other neutralizing antibodies for treatment of 2019-nCoV infections (Tian et al., 2020). Bates et al. 2021 (PMID: 32766589) used CR3022 in a immunofluorescence assay.
-
Beschränkungen
- Nur für Forschungszwecke einsetzbar
-
-
- by
- Akoya Biosciences
- No.
- #104441
- Datum
- 13.09.2022
- Antigen
- SARS-CoV-2 Spike S1
- Chargennummer
- T2023B03
- Validierte Anwendung
- Multiplex Immunohistochemistry
- Positivkontrolle
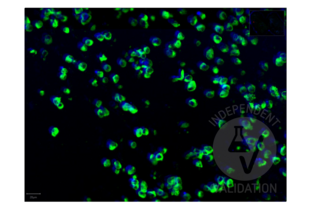
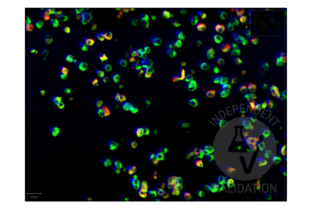
FFPE cell pellets from in vitro cultured human lung cells infected with SARS-CoV-2
- Negativkontrolle
SARS-CoV-2-negative placenta patient sample
- Bewertung
Passed. The anti-SARS-CoV-2 Spike S1 antibody RBD antibody ABIN6952546 produced staining in FFPE cell pellets from in vitro cultured human lung cells infected with SARS-CoV-2.
- Primärantikörper
- ABIN6952546
- Sekundärantikörper
- Full Protocol
- Protocol details are described in the Akoya Biosciences CODEX® User Manual (see https://www.akoyabio.com/wp-content/uploads/2021/01/CODEX-User-Manual.pdf).
- Tissue preparation as outlined in the Akoya Biosciences CODEX® User Manual FFPE tissue protocol.
- Conjugation of the anti-SARS-CoV-2 Spike S1 antibody RBD antibody ABIN6952546 as described in Chapter 4 of the Akoya Biosciences CODEX® User Manual.
- Autofluorescence quenching according to the Autofluorescence Quenching Protocol for CODEX® (see https://www.akoyabio.com/wp-content/uploads/2020/07/Customer-Demonstrated-Protocol-Autofluorescence-Quenching-Mar2020.pdf).
- Anmerkungen
Validierung #104441 (Multiplex Immunohistochemistry)![Erfolgreich validiert 'Independent Validation' Siegel]()
![Erfolgreich validiert 'Independent Validation' Siegel]() ValidierungsbilderProtokoll
ValidierungsbilderProtokoll -
-
Format
- Liquid
-
Konzentration
- 1 mg/mL
-
Buffer
- PBS with 0.02 % Proclin 300.
-
Konservierungsmittel
- ProClin
-
Vorsichtsmaßnahmen
- This product contains ProClin: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
-
Lagerung
- 4 °C,-20 °C
-
Informationen zur Lagerung
- Store at 4°C for up to 3 months. For longer storage, aliquot and store at -20°C.
-
-
-
: "Antibody course and memory B-cell response in the first year after SARS-CoV-2 infection." in: The Journal of infectious diseases, (2022) (PubMed).
: "High antibody levels and reduced cellular response in children up to one year after SARS-CoV-2 infection. ..." in: Nature communications, Vol. 13, Issue 1, pp. 7315, (2022) (PubMed).
: "Safe and effective two-in-one replicon-and-VLP minispike vaccine for COVID-19: Protection of mice after a single immunization." in: PLoS pathogens, Vol. 17, Issue 4, pp. e1009064, (2021) (PubMed).
: "Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody." in: Emerging microbes & infections, Vol. 9, Issue 1, pp. 382-385, (2020) (PubMed).
: "SARS-CoV-2 Seroconversion in Humans: A Detailed Protocol for a Serological Assay, Antigen Production, and Test Setup." in: Current protocols in microbiology, Vol. 57, Issue 1, pp. e100, (2020) (PubMed).
: "A highly conserved cryptic epitope in the receptor-binding domains of SARS-CoV-2 and SARS-CoV." in: Science (New York, N.Y.), (2020) (PubMed).
: "Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody." in: Nature, (2020) (PubMed).
: "Human monoclonal antibody combination against SARS coronavirus: synergy and coverage of escape mutants." in: PLoS medicine, Vol. 3, Issue 7, pp. e237, (2007) (PubMed).
-
-
- SARS-CoV-2 Spike S1
-
Substanzklasse
- Viral Protein
-
Hintergrund
- Spike protein, COVID19, COVID 19, S protein, SARS-CoV S protein, S glycoprotein, E2, Peplomer protein, Spike protein S1, SARS Coronavirus, SARS-CoV-2, SARS CoV 2, 2019-nCoV, Ab1680.10, Ab1680.15, Ab1680.16
-
UniProt
- P59594
Target
-

 (8 Referenzen)
(8 Referenzen) (1 Validierung)
(1 Validierung)



