RNA-DNA Hybrid Antikörper
Kurzübersicht für RNA-DNA Hybrid Antikörper (ABIN4889499)
Target
Reaktivität
Wirt
Klonalität
Applikation
Klon
-
-
Spezifität
- Reacts with RNA-DNA hybrid
-
Aufreinigung
- Purified (protein A)
-
Immunogen
- DNA-RNA duplex (random RNA-DNA heteropolymer)
-
Isotyp
- IgG
-
-
-
Applikationshinweise
-
Working dilution: Optimal dilutions should be determined by the end user.
-
Beschränkungen
- Nur für Forschungszwecke einsetzbar
-
-
- by
- Institute of Photonics and Electronics AS CR, v.v.i.
- No.
- #100000
- Datum
- 23.06.2015
- Antigen
- RNA-DNA Hybrid
- Chargennummer
- Validierte Anwendung
- Surface Plasmon Resonance
- Positivkontrolle
- RNA-DNA Hybrid complexes
- Negativkontrolle
- ssDNA, dsDNA
- Bewertung
The ability of the D5H6 antibody to recognize and bind RNA/DNA hetero-duplexes was approximately twice lower than expected.
- Primärantikörper
- ABIN4889499
- Sekundärantikörper
- Full Protocol
For the experiments a four-channel spectroscopic SPR sensor with a temperature controller developed at IPE was used. In general, the SPR assay was based on the attachment of biotinylated DNA probes to the SPR sensor surface via the streptavidin-biotin interaction, with the streptavidin covalently attached to the alkanthiol self-assembled monolayer, subsequent forming of RNA/DNA hybrid duplexes and monitoring of their interaction with RNA-DNA-hybrid antibody.
The respective steps of the assay were as follows:
- Streptavidin was covalently attached to the sensor surface via amine coupling according to the previously published protocol (Vaisocherová et al. (2006) Biopolymers 82:394-398).
- Biotinylated DNA probes were immobilized in 10mM Tris buffer. The amount of probes was calculated to be of 1012 probes/cm2 and was kept the same across all experiments.
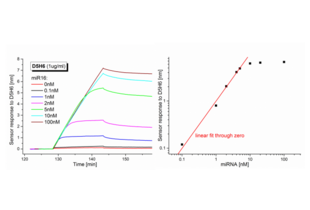
- Complementary miRNA strands were bound in 10mM Tris + 15mM MgCl2 to form RNA-DNA hybrid duplexes. The concentration range of miRNA used was 0.1-100nM.
- The D5H6 at a concentration of 1µg/ml was used in all experiments.
All the experiments were performed at the temperature of 25°C and the flow rate of 20µl/ml.
- Anmerkungen
The sensor response to the binding of D5H6 antibody obtained for various concentrations of miRNA and corresponding calibration curve are shown at Fig.1. The calibration curve is plotted as a dependence of the sensor response to the binding of D5H6 antibody on the concentration of miRNA and is linear up to the miRNA concentration of 5nM.
As a negative control, the ssDNA (only biotinylated DNA probe) or dsDNA (homo-duplex with DNA analogue of the miRNA) surface was used. In both cases, the nonspecific sensor response to the binding of D5H6 was below 2% which falls into the inter-assay variability range.
Validierung #100000 (Surface Plasmon Resonance)![Erfolgreich validiert 'Independent Validation' Siegel]()
![Erfolgreich validiert 'Independent Validation' Siegel]() ValidierungsbilderProtokoll
ValidierungsbilderProtokoll -
-
Format
- Lyophilized
-
Rekonstitution
- Must be reconstituted in distilled water.
-
Konzentration
- 1 mg/mL
-
Buffer
- Tris 0,1M, glycine 0,1M, sucrose 2 %
-
Lagerung
- 4 °C/-20 °C
-
Informationen zur Lagerung
- Lyophilized powder stable for a minimum of 2 years at -20°C. Store reconstituted antibodies at +4°C. For extended periods store in aliquots at -20°C. Antibodies are guaranteed for 6 month from date of receipt.
-
Haltbarkeit
- 24 months
-
-
- RNA-DNA Hybrid
-
Substanzklasse
- Nucleotide
Target
-

 (1 Validierung)
(1 Validierung)



