Leptin ELISA Kits
LEP Reaktivität: Human Colorimetric Sandwich ELISA 0.15 ng/mL - 10 ng/mL Cell Culture Supernatant, Cell Lysate, Plasma, Saliva, Serum, Tissue Homogenate
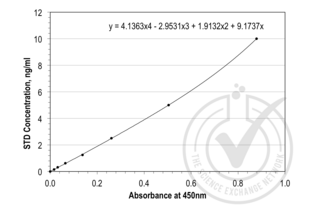
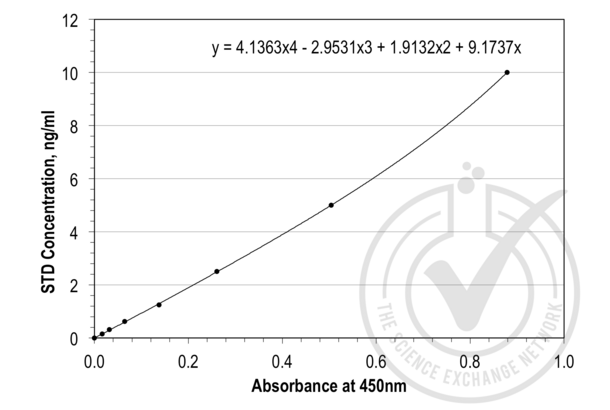 Leptin ELISA Kit (ABIN365165)
Leptin ELISA Kit (ABIN365165)
LEP Reaktivität: Human Colorimetric Sandwich ELISA 0.156-10 ng/mL Plasma, Serum, Tissue Homogenate
LEP Reaktivität: Ratte Colorimetric Sandwich ELISA 0.31 ng/mL - 20 ng/mL Cell Culture Supernatant, Cell Lysate, Plasma, Serum, Tissue Homogenate
Leptin ELISA Kits nach Grade
Hier sind Leptin ELISA Kits mit einem spezifischen Grade zu finden. Die hier aufgeführten Grade sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
Leptin ELISA Kits nach Reaktivität
Hier sind Leptin ELISA Kits für eine Vielzahl von Species wie anti-Human Leptin, anti-Mouse Leptin, anti-Rat Leptin zu finden. Die unten aufgeführten Species gehören zu den verfügbaren Arten. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
Leptin ELISA Kits nach Detektionsmethode
Hier sind Leptin ELISA Kits mit einem spezifischen Detektionsmethode zu finden. Die hier aufgeführten Detektionsmethode sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
Leptin ELISA Kits nach Analytische Methode
Hier sind Leptin ELISA Kits mit einem spezifischen Analytische Methode zu finden. Die hier aufgeführten Analytische Methode sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
Leptin ELISA Kits nach Methodentyp
Hier sind Leptin ELISA Kits mit einem spezifischen Methodentyp zu finden. Die hier aufgeführten Methodentyp sind einige der verfügbaren. Ein Klick auf den entsprechenden Link führt zu den Produkten.
Leptin ELISA Kits nach Bindungsspezifität
Hier sind Leptin ELISA Kits mit einem bestimmten Epitop aufgelistet. Die unten aufgeführten Epitope gehören zu den verfügbaren Epitopen. Klicken Sie auf einen Link, um zu den entsprechenden Produkten zu gelangen.
Häufig verwendete Leptin ELISA Kits
- (9)
- (5)
- (6)
- (1)
- (1)
- (10)
- (1)
- (34)
- (1)
- (1)
- (1)
- (3)
- (1)
- (6)
- (1)
- (3)
- (1)
- (4)
- (1)
- (3)
- (1)
- (2)
- (1)
- (8)
- (1)
- (17)
- (1)
- (1)
- (1)
Aktuelle Publikationen für unsere Leptin ELISA Kits
: "Effect of xylo-oligosaccharides on reproduction, lipid metabolism, and adipokines of hens during the late egg-laying period." in: Animal bioscience, Vol. 35, Issue 11, pp. 1744-1751, (2022) (PubMed).: "Nicotine-mediated upregulation of microRNA-141 expression determines adipokine-intervened insulin resistance." in: Environmental toxicology and pharmacology, Vol. 80, pp. 103506, (2021) (PubMed).
: "Role of TRPV1/TRPV3 channels in olanzapine-induced metabolic alteration: Possible involvement in hypothalamic energy-sensing, appetite regulation, inflammation and mesolimbic pathway." in: Toxicology and applied pharmacology, Vol. 402, pp. 115124, (2021) (PubMed).
: "Brain-Derived Neurotrophic Factor Expression and Signaling in Different Perivascular Adipose Tissue Depots of Patients With Coronary Artery Disease." in: Journal of the American Heart Association, Vol. 10, Issue 6, pp. e018322, (2021) (PubMed).
: "Puberty is a critical window for the impact of diet on mammary gland development in the rabbit." in: Developmental dynamics : an official publication of the American Association of Anatomists, Vol. 248, Issue 10, pp. 948-960, (2020) (PubMed).
: "Paternal chronic folate supplementation induced the transgenerational inheritance of acquired developmental and metabolic changes in chickens." in: Proceedings. Biological sciences, Vol. 286, Issue 1910, pp. 20191653, (2020) (PubMed).
: "Dietary supplementation of Morus nigra L. leaves decrease fat mass partially through elevating leptin-stimulated lipolysis in pig model." in: Journal of ethnopharmacology, Vol. 249, pp. 112416, (2020) (PubMed).
: "Serum Asprosin Concentrations Are Increased and Associated with Insulin Resistance in Children with Obesity." in: Annals of nutrition & metabolism, Vol. 75, Issue 4, pp. 205-212, (2020) (PubMed).
: "Upregulation of MC4R and PPAR-α expression mediates the anti-obesity activity of Moringa oleifera Lam. in high-fat diet-induced obesity in rats." in: Journal of ethnopharmacology, Vol. 251, pp. 112541, (2020) (PubMed).
: "Prenylated Phenolic Compounds from the Aerial Parts of Glycyrrhiza uralensis as PTP1B and α-Glucosidase Inhibitors." in: Journal of natural products, Vol. 83, Issue 4, pp. 814-824, (2020) (PubMed).
Aliase für Leptin ELISA Kits
leptin (Lep) ELISA Kitsleptin (LEP) ELISA Kits
leptin (lep) ELISA Kits
LEPD ELISA Kits
ob ELISA Kits
OB ELISA Kits
obese ELISA Kits
OBS ELISA Kits
Haben Sie etwas anderes gesucht?
- LEPREL1 ELISA Kits
- Leprecan-Like 4 ELISA Kits
- LEPRE1 ELISA Kits
- LEMD3 ELISA Kits
- Leiomodin 1 ELISA Kits
- LEFTY2 ELISA Kits
- LEFTY1 ELISA Kits
- LEF1 ELISA Kits
- Lectin, Galactoside-Binding, Soluble, 3 Binding Protein ELISA Kits
- Lectin, Galactoside-Binding, Soluble, 2 ELISA Kits
- LECT2 ELISA Kits
- LECT1 ELISA Kits
- LEAP2 ELISA Kits
- LDLRAP1 ELISA Kits
- LDL ELISA Kits
- LDHD ELISA Kits
- LDHB ELISA Kits
- LCT ELISA Kits
- LCP2 ELISA Kits
- LCP1 ELISA Kits
- Leptin Receptor ELISA Kits
- LETMD1 ELISA Kits
- Leucine-Rich, Glioma Inactivated 1 ELISA Kits
- Leukotriene B4 Receptor/BLT ELISA Kits
- Leupaxin ELISA Kits
- LFNG ELISA Kits
- LGALS1/Galectin 1 ELISA Kits
- LGALS12 ELISA Kits
- LGALS13 ELISA Kits
- LGALS14 ELISA Kits
- LGALS7 ELISA Kits
- LGALS8 ELISA Kits
- LGALS9 ELISA Kits
- LGI3 ELISA Kits
- LGMN ELISA Kits
- LGR4 ELISA Kits
- LGR5 ELISA Kits
- LHB ELISA Kits
- LHCGR ELISA Kits
- LHFPL5 ELISA Kits




